GalNAc Application in Oligonucleotide Delivery
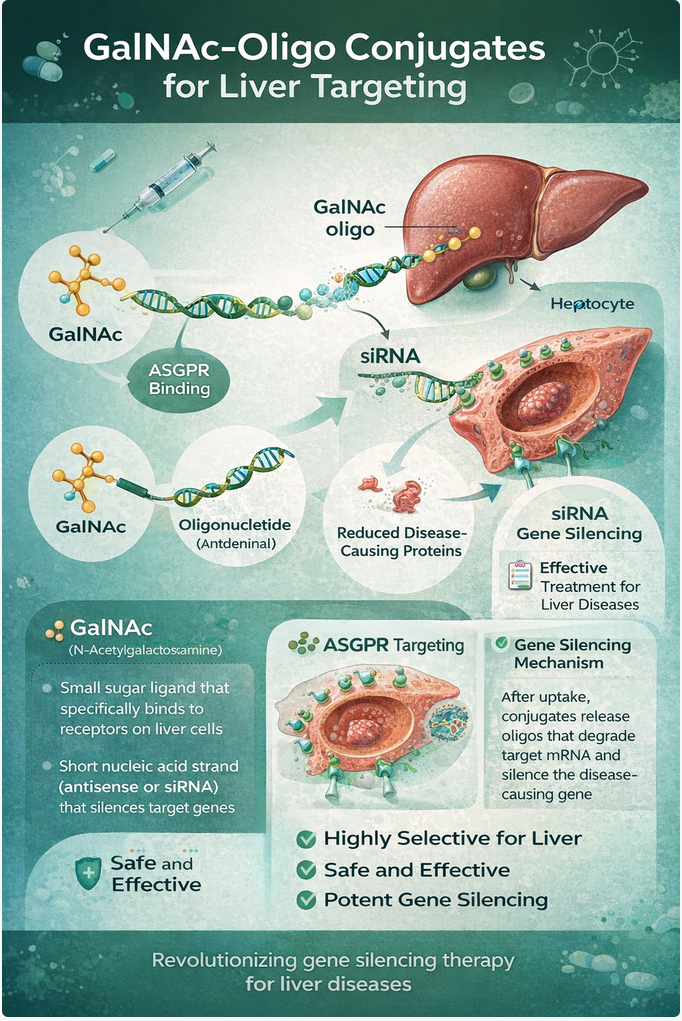
The emergence of RNA-based therapeutics, particularly small interfering RNA (siRNA) and antisense oligonucleotides (ASOs), has demonstrated significant potential for treating various illnesses. However, their clinical application has been hampered by challenges such as inefficient cellular uptake, off-target toxicity, and rapid clearance from the body. One of the most effective solutions to these issues is N-acetylgalactosamine (GalNAc) conjugation, a proven method for delivering oligonucleotide therapies specifically to the liver. This method utilizes the asialoglycoprotein receptor (ASGPR), which is abundant on liver cells and contains over 500,000 receptors per cell. GalNAc ligands, particularly those containing tri-GalNAc clusters, bind to ASGPR with high affinity. This strong binding promotes rapid receptor-mediated endocytosis, enabling the efficient internalization of the GalNAc-oligonucleotide conjugate.
From a manufacturing and design perspective, GalNAc-conjugated systems bring significant benefits. The delivery method is chemically well-defined, consisting of a small-molecule ligand covalently linked to the oligonucleotide, which simplifies CMC endeavors, as well as regulatory oversight. Moreover, this approach is versatile, compatible with both siRNA and ASO formats, and can be incorporated through solid-phase synthesis or post-synthetic conjugation without compromising oligonucleotide stability. Clinically, the implementation of GalNAc delivery has led to the successful development of multiple GalNAc-siRNA therapeutics that demonstrate substantial and durable target knockdown with minimal dosing requirements, resulting in favorable safety profiles and the practicality of subcutaneous administration. The proven success of these agents highlights GalNAc conjugation as a platform technology, one that fundamentally redefines the treatment landscape for oligonucleotide therapeutics, moving beyond a mere solution for specific products.
Despite its advancements, GalNAc delivery does encounter certain limitations. A significant challenge is that only a small percentage of the internalized oligonucleotide escapes the endosomal compartment to reach the cytosol or nucleus, underscoring endosomal escape as a critical biological bottleneck. Additionally, ASGPR expression is predominantly confined to hepatocytes, limiting the applicability of GalNAc to liver-targeted therapies. Current research is aimed at enhancing intracellular trafficking efficiencies, refining linker chemistry, and discovering analogous ligand-receptor systems for targeting beyond the liver. In conclusion, GalNAc conjugation signifies a pivotal development in oligonucleotide delivery, merging precise biological targeting with scalable chemistry and consistently demonstrating clinical validation. While ongoing innovations are set to address endosomal escape and broaden the therapeutic reach, GalNAc remains the benchmark for hepatocyte-directed oligonucleotide therapeutics and future delivery platforms.
Nair JK, et al. J Am Chem Soc. 2014 Dec 10;136(49):16958-61.
Springer AD, Dowdy SF. Nucleic Acid Ther. 2018 Jun;28(3):109-118.
Brown CR, et al. Nucleic Acids Res. 2020 Dec 2;48(21):11827-11844.