Shifting Paradigms: The Impact of Tumor Biology on Antibody-Drug Conjugate (ADC) Development
The domain of antibody-drug conjugates (ADCs) has made significant investments in enhancing linkers, payloads, and conjugation chemistry. Traditionally, attention has focused on improving linker stability, optimizing drug-to-antibody ratios (DAR), developing novel payload classes, and achieving site-specific conjugation. However, contemporary challenges are increasingly revolving around factors such as target density and heterogeneity, tumor penetration, intracellular sensitivity, and adaptive resistance. This evolution is prompting a shift in focus from chemistry-first to a biology-first approach to innovation.
Recent advancements are emerging from an enriched understanding of target biology, biomarker-driven patient selection, and the investigation of novel biological mechanisms, including immune-stimulating ADCs, non-internalizing ADCs, and conditionally activated payloads. Consequently, the trajectory of ADC technology development is becoming less constrained by technological limitations and more influenced by the complexities inherent to tumor biology. Future breakthroughs are anticipated to rely increasingly on insights into biological processes rather than mere incremental enhancements in linker or payload design.
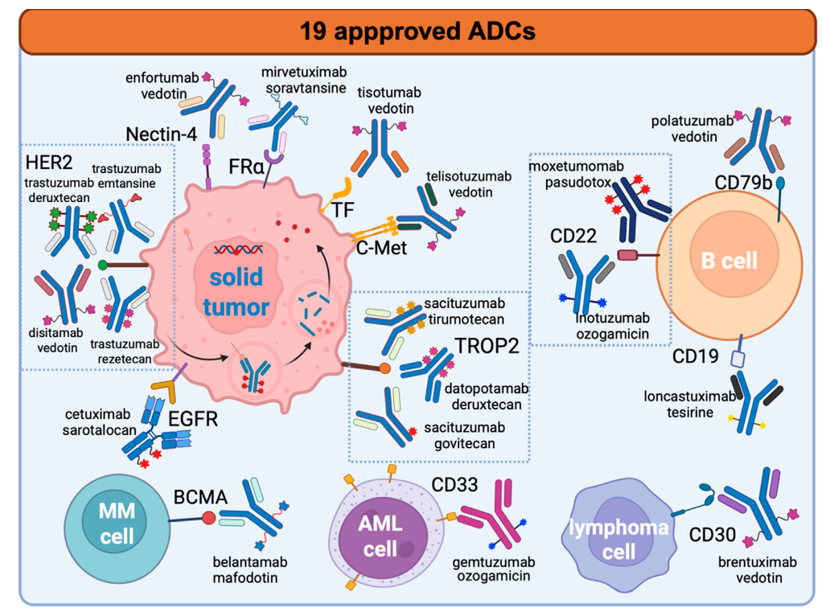
The overview of the 19 approved antibody-drug conjugates (ADCs) highlights the crucial role of biological factors in the development of ADC therapies. Clinical success is most often observed in environments with highly favorable biological targets, particularly in hematologic malignancies (such as CD19, CD22, CD30, CD33, and BCMA). These conditions are characterized by strong, uniform antigen expression, effective internalization mechanisms, and minimal barriers to physical delivery. In contrast, the effectiveness of ADCs in solid tumors (including those targeting HER2, TROP2, Nectin-4, FRα, TF, c-Met, and EGFR) is limited to specific situations where antigen density, trafficking, and payload sensitivity are optimally matched. As a result, treatment responses in these instances are often more variable and less durable. This reinforces the idea that tumor biology, rather than chemical properties, is the primary constraint on the future growth of ADC therapies.